
This method of ATP generation is called "substrate level phosphorylation" and it occurs in glycolysis which you studied as the mechanism for ATP production in animal cells that do not contain mitochondria. The best example of such a cell type is the erythrocyte. Cells that do contain mitochondria also process glucose by this metabolic pathway and they derive a tiny proportion of their ATP by this method.
This method of ATP production depends on phosphorylated intermediary metabolites which have a high "phosphoryl group transfer potential" and kinases can use them directly as phosphoryl group donors for ATP synthesis. Substrates which yield ATP by this method are described as being metabolised by fermentation.
Glycolysis is an example of fermentation and so it is accurate to say that erythrocytes carry out fermentation of glucose even though this is not how erythrocyte metabolism is usually described in biochemistry text books.
The diagram below is on overview of glycolysis; showing the coupled redox processes which maintain the cell's redox balance and the kinase catalysed steps which generate ATP.

Glycolysis is a metabolic process that yields ATP. You should note that an oxidation step, using NAD as the oxidizing agent, occurs in this metabolic sequence. Molecular oxygen is not used anywhere in this process and this is why glycolysis is described as an anaerobic process. Glycolysis can in fact occur in the presence of molecular oxygen, as is seen in the erythrocyte, but oxygen is not needed for this process and can not be used even if it is present.
You will see that in this scheme lactate is the product of this route for the anaerobic use of glucose for ATP synthesis. This is necessary because of the need for the cell to maintain redox balance. Under anaerobic conditions redox balance is maintained in the cell by coupling the use of pyruvate, as an oxidising agent, to provide for NAD regeneration which results in the production of lactate. Lactate is a waste product and is released from the cell. It's production is necessitated by having to use pyruvate as a dump for reducing equivalents to allow for regeneration of oxidised NAD.
In many heterotrophic species of microorganisms that live under anaerobic conditions glycolysis is the primary route for ATP synthesis.
As we have noted above glycolysis coupled with lactate production is a simple example of fermentation.
Other examples of fermentation, with products other than lactate, occur in rumen bacteria and these are dealt with in detail later in this teaching unit.
Animal cells containing mitochondria carry out aerobic respiration and use oxygen as the dump for reducing equivalents. Under these circumstances pyruvate doesn't have to be used as an oxidising agent. It can itself be oxidised to acetyl CoA, and ultimately to CO2, via the TCA cycle. The redox reactions of the TCA cycle supply reducing equivalents for oxidative phosphorylation whose final redox reaction is the reduction of oxygen to form water. Oxidative phosphorylation generates a much higher yield of ATP from glucose as the starting substrate, than is possible in the anaerobic fermentation pathway that occurs in the erythrocyte.
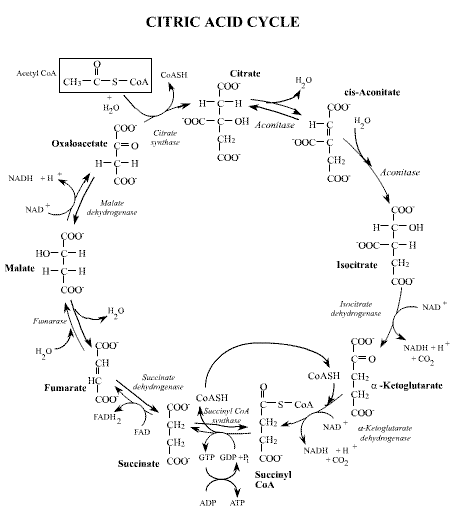
The diagram below shows the essential elements of the Tricarboxylic acid cycle.
The cycle occurs within mitochondria found in eukaryote cells and redox reactions generate reduced intermediary electron carriers which are reoxidized in the process of oxidative phosphorylation which generates the bulk of ATP in these cells. These processes working together constitute aerobic respiration.
As we study patterns of anerobic metabolism in the microorganisms that live in the reticulo-rumen we shall see instances of these reactions operating in the reverse direction to subserve the objective of regenerating NAD.
Saccharomyces fermentation
A slightly more complex mechanism than lactate production for achieving NAD regeneration, is seen in Saccharomyces as shown in the next scheme.
The reactions that are different from that in the erythrocyte are:

This fermentation is more complex than that seen in the erythrocyte. The conversion of pyruvate to the final products ethanol and CO2 requires two steps and uses two enzymes not present in the erythrocyte. This yeast is used in the brewing and baking industries for its ethanol and CO2 generating properties.
In the reticulo-rumen there are many species of micro-organisms that carry out carbohydrate fermentation using different enzymes to those found in yeast or in the erythrocyte. They have fermentation end products much more diverse than lactate or ethanol and carbon dioxide, and many of the fermentation products are valuable nutrients for the host ruminant.
We study these metabolic pathways later in this CAL.